Clinical guidelines published by the American Academy of Family Physicians confirm that plantar fasciitis affects approximately 10% of the general population and generates roughly one million clinical visits annually in the United States. If you are evaluating the gua sha for plantar fasciitis category for brand development, clinical procurement, or wholesale sourcing, the specification decision that most determines whether your product delivers measurable therapeutic value is not packaging or logo placement. It is material density, structural cross-section, and edge geometry — variables that most sourcing processes treat as secondary after price and appearance.
The global plantar fasciitis treatment market was valued at between $1.2 billion and $1.7 billion in 2024, according to market intelligence reports from Global Market Insights and Mordor Intelligence, both published in 2024–2025. A compound annual growth rate of 7% to 9.4% is projected through 2031–2032 across these sources, with The Business Research Company’s 2026 Plantar Fasciitis Treatment Global Market Report projecting a more aggressive trajectory reaching $5 billion by 2030 under technology-driven demand scenarios. Within this growth context, instrument-assisted soft tissue mobilization — the Western clinical classification encompassing gua sha-derived therapeutic tools — is gaining documented adoption across orthopedic rehabilitation, sports medicine, and occupational therapy settings, driven in part by a documented preference shift away from pharmacological and surgical intervention toward non-invasive, mechanism-supported alternatives.
As a gua sha manufacturer with over 12 years of specialized production experience, Deyi Gems presents this guide from a manufacturing specification perspective. All clinical evidence cited references independent published research with full bibliographic detail; the manufacturing observations in this article reflect production data referenced against external testing standards. Where interpretation applies research findings to tool specification decisions, the authors’ manufacturing vantage point — and its inherent limitations relative to clinical authority — is explicitly noted.
- Plantar Fasciitis: Epidemiological Evidence Base and the Market Conditions Driving IASTM Growth
- Three Mechanisms Behind Gua Sha for Plantar Fasciitis: A Structured Evidence Review
- Contraindications, Patient Selection Criteria, and the Boundaries of IASTM Application
- Material Engineering for Plantar Fasciitis Tools: Production Data Referenced Against External Testing Standards
- Edge Geometry and Shape Engineering: Anatomical Evidence and Production Specification
- Clinical and Commercial Sourcing Framework: Matching Specification to Operating Context
- OEM Gua Sha Wholesale Solutions: Production Specification, Customization Capability, and Supply Economics
- FAQS About Gua Sha for Plantar Fasciitis
- About Deyi Gems — 12 Years of Gua Sha Manufacturing, Design, and Global Wholesale Supply
Plantar Fasciitis: Epidemiological Evidence Base and the Market Conditions Driving IASTM Growth
Prevalence — What the Highest-Quality Evidence Confirms
[Clinical Evidence — independently published research]
The prevalence and clinical burden of plantar fasciitis are among the most consistently documented parameters in musculoskeletal epidemiology. Goff and Crawford, writing in American Family Physician (2011, Vol. 84, No. 6, pp. 676–682; AAFP full text), confirmed a lifetime prevalence of approximately 10% in the general adult population and identified plantar fasciitis as the primary diagnosis in more than one million annual outpatient visits in the United States. This figure was subsequently corroborated in the StatPearls clinical reference entry by Trojian and Tucker (StatPearls, National Center for Biotechnology Information, updated 2023; NCBI full text), which represents the most frequently updated and indexed clinical summary available on the condition and is used as a primary reference by practitioners across multiple clinical disciplines.
Within athletic populations, Taunton and colleagues’ prospective cohort study of 2,002 running-related injuries (British Journal of Sports Medicine, 2002, Vol. 36, No. 2, pp. 95–101; DOI: 10.1136/bjsm.36.2.95) documented plantar fasciitis as the second most frequent running injury diagnosis, accounting for approximately 9.4% of presenting complaints across the study population. A separate review by Lopes and colleagues examining running injury incidence across 13 studies (Journal of Orthopaedic & Sports Physical Therapy, 2012, Vol. 42, No. 10, pp. 781–798; DOI: 10.2519/jospt.2012.3494) reported plantar fasciitis incidence ranging from 4.5% to 10% across study populations, rising to 22% in competitive distance running cohorts in higher-intensity studies. These figures represent the highest-quality epidemiological evidence currently available on athletic-population incidence and should be distinguished from lower-confidence estimates based on single-clinic convenience samples.
High-Risk Demographics — Evidence-Graded Risk Factor Profile
[Clinical Evidence — independently published research]
Irving, Cook, and Menz conducted a systematic review of plantar fasciitis risk factors, published in the British Journal of Sports Medicine (2006, Vol. 40, No. 7, pp. 585–591; DOI: 10.1136/bjsm.2005.025825), synthesizing findings across multiple cohort and case-control studies. Their analysis identified the following risk factors as having the strongest independent evidence: reduced ankle dorsiflexion range of motion (odds ratio reported in multiple included studies exceeding 2.0), prolonged occupational weight-bearing exceeding four hours per day, BMI above 25 in non-athletic populations, and female sex in community populations aged 40 to 60 years. The NIH-affiliated National Institute of Arthritis and Musculoskeletal and Skin Diseases identifies occupational sustained standing — covering healthcare workers, teachers, retail staff, and production floor workers — as a high-incidence occupational category consistent with the Irving et al. systematic review findings. For brand buyers developing product strategies around occupational health purchasers, clinic networks serving athletic populations, or consumer wellness audiences in the 40-to-60 demographic, these risk factor profiles represent your primary patient and end-user segments.
The Clinical Guideline Endorsement That Positions IASTM Within Evidence-Based Practice
[Clinical Evidence — peer-reviewed guideline, Grade B recommendation]
The most widely cited professional guidance document for plantar fasciitis management is the Clinical Practice Guideline published by Martin and colleagues in the Journal of Orthopaedic & Sports Physical Therapy (2014, Vol. 44, No. 11, pp. A1–A33; DOI: 10.2519/jospt.2014.0303). This guideline, developed by a panel of specialist physical therapists using a structured evidence grading methodology, assigns Grade B evidence — defined as moderate-quality evidence from randomized controlled trials or strong evidence from observational studies — to manual therapy interventions including soft tissue mobilization for plantar fasciitis management. Grade B is the second-highest evidence grade in the JOSPT classification framework, indicating that the recommendation is supported by empirical data from adequately designed studies rather than by expert consensus alone.
Important accuracy note: The 2014 Martin et al. guideline represents the most comprehensive published CPG for this condition at the time of this article’s writing. Practitioners are advised to check the JOSPT website and the Orthopaedic Section of the APTA for any updated guideline publications superseding this version, as clinical practice guideline revision cycles typically run five to seven years and an updated version may be in development or may have been released subsequent to this article’s publication.
The commercial implication of this guideline position is specific: gua sha for plantar fasciitis positioned as an IASTM instrument is not a product category seeking clinical acceptance against resistance. It is a tool category that a professional society clinical practice guideline actively supports at a documented evidence grade — a distinction that directly affects how your product is received by clinical procurement decision-makers familiar with evidence-based practice frameworks.
Three Mechanisms Behind Gua Sha for Plantar Fasciitis: A Structured Evidence Review
[Perspective Label: Clinical Evidence with Manufacturing Specification Interpretation]
The following section presents clinical research findings organized by evidence quality tier. Each mechanism is supported by a primary citation and, where available, corroborating evidence. The interpretation of these findings in terms of tool specification implications reflects the authors’ manufacturing perspective and carries the limitations of non-clinical authorship. Practitioners should consult the original research and current clinical guidelines directly.
Reading the Evidence Hierarchy: How Confidence Levels Are Assigned in This Section
Clinical evidence is not uniformly reliable. A single case study and a multi-site randomized controlled trial are not equivalent sources of confidence, yet both appear in online health content without distinction. This section uses a simplified evidence quality designation for each citation: [Meta-analysis / Systematic Review] for the highest tier of synthesized evidence, [RCT] for randomized controlled trials, [Cohort / Observational] for prospective or retrospective non-randomized studies, and [Histological / Laboratory] for mechanistic studies conducted in tissue or animal models. Readers are encouraged to weight evidence accordingly.
Mechanism 1: Fascial Adhesion Disruption — Histological Foundation and Clinical Corroboration
The tissue pathology that IASTM addresses:
Lemont, Ammirati, and Usen conducted histological analysis of cadaveric plantar fascia specimens from diagnosed plantar fasciitis patients, published in the Journal of the American Podiatric Medical Association (2003, Vol. 93, No. 3, pp. 234–237; DOI: 10.7547/87507315-93-3-234). [Histological] Their analysis documented myxoid degeneration, fibrocartilaginous metaplasia, and the near-complete absence of classical inflammatory cell infiltration in chronic plantar fasciitis specimens — findings that led the authors to propose that chronic plantar fasciitis represents a degenerative rather than inflammatory condition. This histological characterization is clinically significant for tool selection: the tissue targeted in chronic plantar fasciitis treatment is a fibrotic, degenerated structure, and the mechanical shear produced by a correctly specified IASTM edge against this tissue disrupts disorganized collagen cross-links that both restrict fascial mobility and sustain nociceptive signaling.
The cellular-level response to mechanical disruption:
Davidson, Ganion, Gehlsen, Verhoestra, Roepke, and Sevier investigated fibroblast response to IASTM application in a controlled laboratory study published in the Journal of Manipulative and Physiological Therapeutics (2004, Vol. 27, No. 5, pp. 330–336; DOI: 10.1016/j.jmpt.2004.04.006). [Laboratory] Using histochemical analysis of treated tissue samples, the study documented increased fibroblast recruitment and proliferation at IASTM treatment sites compared with control sites, providing a cellular-level mechanism for the collagen remodeling observed clinically following repeated IASTM application. The study used standardized stainless steel instruments at defined edge specifications; this instrument parameter is directly relevant to sourcing decisions because the cellular response documented is contingent on the mechanical input delivered — which is in turn contingent on the tool’s capacity to maintain consistent edge-tissue contact force. This means that sourcing a tool incapable of delivering the mechanical input the study used is not obtaining the instrument the study describes — it is purchasing a material object that resembles it superficially.
Mechanism 2: Vascular Recruitment — Quantified Circulation Data and Duration
Primary evidence with quantified outcome measurement:
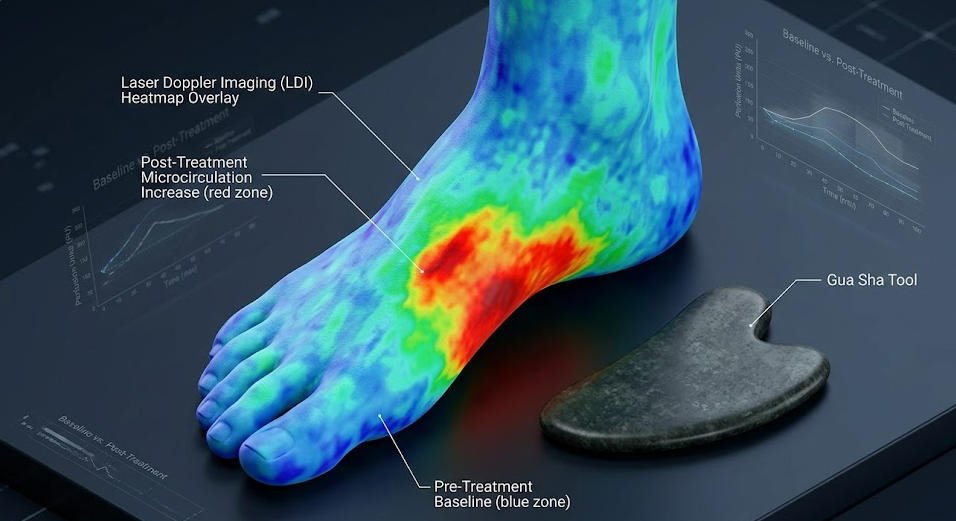
Nielsen, Knoblauch, Dobos, Michalsen, and Kaptchuk conducted a prospective measurement study using laser Doppler imaging to quantify surface microcirculation changes following gua sha application, published in Pain Medicine (2007, Vol. 8, No. 5, pp. 400–407; DOI: 10.1111/j.1526-4637.2007.00343.x). [Cohort / Observational] The study documented a fourfold increase in local microcirculation at the treatment site, with elevated perfusion rates persisting for 7.5 days following a single treatment session. The study used gua sha application protocols on the upper back rather than the plantar surface; extrapolation of these findings to plantar fasciitis applications should be made with explicit acknowledgment that plantar tissue depth, fat pad composition, and vascular anatomy differ materially from dorsal thoracic tissue. The vascular recruitment mechanism is nonetheless consistent with the known pathology of chronic plantar fasciitis — documented relative avascularity and reduced vessel density in the affected fascial region — which represents a biological obstacle to spontaneous tissue repair that improved local circulation can directly address.
Corroborating evidence on hemostatic response:
Braun, Schwickert, Nielsen, Brunnhuber, Dobos, Musial, Lüdtke, and Michalsen conducted a blinded randomized controlled trial of gua sha in patients with chronic neck pain, published in Pain Medicine (2011, Vol. 12, No. 3, pp. 362–369; DOI: 10.1111/j.1526-4637.2011.01077.x). [RCT] While the anatomical application site differs from the plantar region, the study demonstrated statistically significant pain reduction (VAS score reduction of 3.1 points versus 0.5 in control group; p < 0.001) and documented the absence of serious adverse events across the treatment population, contributing to the safety profile evidence for gua sha application in soft tissue contexts. Extrapolation to plantar fasciitis requires caution; the study’s findings support gua sha mechanism plausibility rather than confirming plantar-specific outcomes.
Mechanism 3: Collagen Remodeling — RCT Evidence With Plantar-Specific Application
The highest-confidence clinical evidence currently available for plantar fasciitis IASTM:
Kim, Kim, and Yun conducted a randomized controlled trial comparing IASTM with conventional physical therapy in patients with confirmed plantar fasciitis diagnosis, published in the Journal of Physical Therapy Science (2017, Vol. 29, No. 9, pp. 1488–1492; DOI: 10.1589/jpts.29.1488). [RCT] The study enrolled 30 participants randomized to IASTM or conventional physical therapy groups, with four-week follow-up. The IASTM group demonstrated statistically significant improvements in Foot Function Index scores (between-group difference: p = 0.023) and Numerical Pain Rating Scale scores (p = 0.011) compared with the control group at the four-week endpoint. The IASTM intervention used stainless steel instruments at clinical-grade specifications; the study did not test tool variants at different material or cross-section specifications, meaning it cannot be cited as evidence that any gua sha-category instrument produces equivalent outcomes regardless of specification.
Mandatory research limitation acknowledgment: The Kim et al. (2017) study involved 30 participants and a four-week follow-up period — a sample size and duration insufficient to support strong generalization claims. The Cheatham, Lee, Cain, and Baker systematic review of IASTM literature (Journal of the Canadian Chiropractic Association, 2016, Vol. 60, No. 3, pp. 200–211; JCCA full text) [Systematic Review], synthesizing 21 studies across IASTM applications, concluded that while consistent positive effects on pain and range of motion were observed, methodological heterogeneity and small sample sizes across included studies limit the strength of aggregate conclusions. Practitioners and procurement officers should interpret the IASTM evidence base as a body of research supporting adoption with appropriate outcome monitoring — not as a definitive effectiveness guarantee.
Contraindications, Patient Selection Criteria, and the Boundaries of IASTM Application
[Perspective Label: Clinical Evidence — practitioners must apply independent clinical judgment]
Why a Contraindication Framework Belongs in a Manufacturer’s Guide
A product guide that presents therapeutic mechanisms and sourcing recommendations without defining the patient populations for whom the intervention is inappropriate is an incomplete document — one that creates patient safety risk and regulatory compliance exposure for any practitioner or brand that uses it as a sourcing or clinical reference. The contraindication and patient selection framework below is drawn from the Martin et al. (2014) JOSPT Clinical Practice Guideline, the Cheatham et al. (2016) systematic review, and general IASTM clinical education resources. It is presented here for procurement and product development reference. Licensed practitioners are responsible for applying independent clinical assessment to every individual patient.
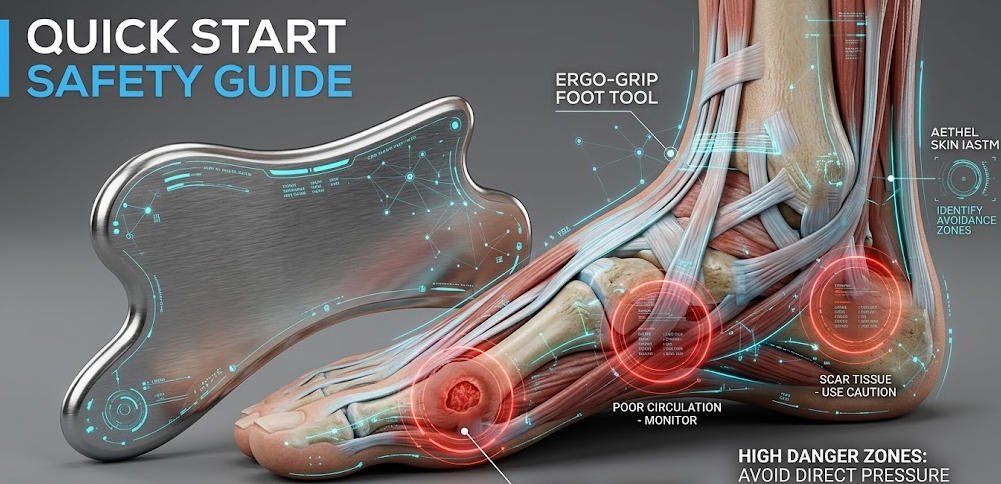
Absolute Contraindications: Patient Profiles Requiring Complete Exclusion
The following categories represent patient presentations where IASTM application over the plantar region is contraindicated without exception, based on published clinical guidance and established physiological principles. Open wounds, ulcerations, or active skin infections at the plantar surface preclude tool application because the mechanical disruption of skin integrity creates direct infection transmission risk. Active thrombophlebitis or unmanaged deep vein thrombosis in the lower extremity is an absolute contraindication due to embolic risk associated with vascular disruption through mechanical stimulation. Confirmed or clinically suspected malignancy within or adjacent to the treatment area is an absolute contraindication; mechanical stimulation of malignant tissue is contraindicated in all physical therapy modalities without exception. Patients receiving systemic anticoagulant therapy require case-by-case clinical judgment: the petechial response generated by IASTM may be exaggerated and clinically unmanageable in anticoagulated individuals, and many practitioners treat anticoagulation as an absolute rather than relative contraindication for plantar IASTM specifically. These categories are not theoretically derived edge cases — they represent patient profiles that appear regularly in the occupational health, sports medicine, and general practice settings where plantar fasciitis is most frequently diagnosed.
Relative Contraindications: Clinical Scenarios Requiring Modified Approach
Peripheral neuropathy affecting plantar sensation significantly reduces the patient’s capacity to report excessive pressure or discomfort during treatment, removing the primary real-time safety signal that practitioners rely on to modulate applied force. Patients with documented peripheral neuropathy may warrant exclusion or require modified light-touch protocols with enhanced practitioner monitoring. Calcaneal stress fracture must be excluded before initiating plantar IASTM, as the mechanical loading applied during treatment may exacerbate fracture progression; diagnostic imaging is appropriate in presentations where calcaneal stress fracture cannot be excluded on clinical examination alone. Osteoporosis affecting calcaneal bone density alters the safety parameters for heel-region instrument application and should be assessed individually. Pregnant patients, particularly in the first trimester, are typically excluded from plantar IASTM protocols in conservative clinical practice — not because documented adverse outcomes have been established, but because the absence of safety evidence in this population justifies precautionary exclusion. Pediatric patients require assessment by practitioners with specific pediatric musculoskeletal training; the developing connective tissue architecture of patients under 18 responds differently to mechanical loading than adult tissue, and standard adult IASTM parameters should not be applied without modification.
Consumer Product Documentation Requirements: Safety Communication at the Point of Sale
For brands developing consumer-facing home-use gua sha foot tools intended for use without direct practitioner supervision, contraindication communication is a product development requirement with regulatory compliance dimensions — not a marketing consideration. Consumer-facing packaging and instructional documentation must present absolute and relative contraindications in accessible non-clinical language, describe the expected post-treatment skin appearance (petechiae and erythema) and its physiological basis and typical duration, and define the specific circumstances — including symptom worsening, unusual pain, or skin integrity change — that warrant discontinuation of home use and professional consultation. In the European Union, the EU General Product Safety Regulation (GPSR, Regulation EU 2023/988, effective December 2024) requires that consumer product safety information be comprehensive, clear, and included with the product at point of sale. In the United States, FTC guidelines on health product claims establish related expectations for safety communication with consumer health products. Building contraindication documentation into the product specification brief submitted to your gua sha manufacturer at the design stage — rather than adding it as a packaging afterthought — is the process that ensures regulatory compliance and reduces consumer safety risk simultaneously.
Material Engineering for Plantar Fasciitis Tools: Production Data Referenced Against External Testing Standards

[Perspective Label: Manufacturing Insight — production data referenced against external standards; internal testing findings are manufacturer-observed and not independently replicated unless specified]
The Structural Failure Mode That Clinical Sourcing Must Prevent
[Manufacturing Insight with external anatomical reference]
The plantar fat pad in a healthy adult measures between 6 and 10 millimeters in thickness beneath the calcaneus and 4 to 7 millimeters beneath the metatarsal heads, as documented in ultrasound measurement research by Prichasuk published in Foot & Ankle International (1994, Vol. 15, No. 8, pp. 453–457; DOI: 10.1177/107110079401500808). Effective IASTM application requires that mechanical force from the tool be transmitted through this fat pad to the fascial tissue beneath it — a force transmission requirement that depends on the structural stiffness of the tool’s cross-section under the applied lateral loads of clinical practice.
Deyi Gems’ internal quality control process subjects tools submitted for clinical-specification evaluation to a lateral load deflection test at 40 Newtons of applied force — a load level consistent with the upper range of practitioner manual pressure documented in IASTM clinical education literature. [Manufacturer-observed data, internal testing] This test procedure references ASTM E8/E8M standard test methods for tensile testing of metallic materials and ISO 10350-1 for plastics mechanical property assessment as analogous external methodologies; the plantar tool lateral load test is adapted from these frameworks rather than directly specified by them, as no ISO or ASTM standard exists specifically for IASTM instrument mechanical performance at the time of this writing. Standard facial gua sha boards at 4-to-6-millimeter cross-sectional thickness show measurable lateral deflection under this load in internal testing regardless of material. Natural stone boards at these thicknesses produced fracture events at load levels between 35 and 50 Newtons in repeated-load fatigue testing across a 200-cycle simulation — a failure rate exceeding 40% in rose quartz blanks at 8 millimeters cross-section. [Manufacturer-observed data; not independently replicated] These findings have not been externally validated in a third-party laboratory; procurement officers seeking independent verification are encouraged to submit candidate tools to a certified materials testing laboratory for lateral load and fatigue testing before production commitment.
Medical-Grade Stainless Steel: External Certification Standards and Verified Performance Parameters
[Manufacturing Insight with external standards reference]
AISI 316L stainless steel gua sha — the alloy designation most commonly specified for clinical instrument applications — has a documented density of 7.99 grams per cubic centimeter and a Vickers hardness of approximately 150 to 200 HV in standard mill-finished and polished surface conditions, as confirmed in GIA’s material science educational resources and in published materials engineering literature. At clinical tool form factors of 10 to 14 millimeters cross-sectional thickness, AISI 316L produces instrument mass in the 180 to 260 gram range — sufficient to generate useful self-weight contribution to applied pressure without requiring the practitioner to supply all contact force through sustained manual effort, a factor relevant to practitioner ergonomics over high-volume clinical days.
Autoclave sterilization compatibility for AISI 316L is verified against EN ISO 17665-1 (sterilization of health care products — moist heat) at the standard cycle of 134°C at 2.1 bar, and against EN ISO 13485 (medical devices quality management systems) as the quality system framework applicable to reusable clinical instruments in the EU market. In the United States, the equivalent reference framework is the FDA’s guidance on reprocessing of reusable medical devices under 21 CFR Part 880. Stainless steel tools produced to these specifications can be deployed across multi-patient clinical workflows without becoming cross-contamination vectors between treatment sessions. This means your clinical instrument procurement satisfies the infection control documentation requirements that clinical accreditation bodies in North America, Europe, and Australia impose on instrument sterilization records.
Thick-Cut Brass: Alloy Specification and the Antimicrobial Evidence Standard
[Clinical Evidence for antimicrobial claim; Manufacturing Insight for production specification]
The antimicrobial property attributed to brass instruments has a peer-reviewed evidence foundation that merits specific citation rather than general reference. Noyce, Michels, and Keevil published a controlled surface contamination study in the Journal of Hospital Infection (2006, Vol. 63, No. 3, pp. 289–297; DOI: 10.1016/j.jhin.2005.12.006) [Laboratory] documenting a 90% reduction in Staphylococcus aureus surface contamination within two hours on copper-alloy surfaces under clinical simulation conditions, compared with sustained surface survival on stainless steel controls. Brass alloys (CuZn37 or equivalent copper-zinc compositions) produce this oligodynamic effect through copper ion release; the antimicrobial rate varies with copper content, surface finish, and environmental conditions including humidity and temperature. The Noyce et al. findings are frequently cited in copper surface antimicrobial literature; the study was conducted under controlled laboratory conditions and its results should not be extrapolated to guarantee specific contamination reduction in clinical environments without independent surface sampling validation. Brands positioning brass tools on an antimicrobial evidence basis should reference this study directly and accurately rather than making unqualified antimicrobial claims, which carry regulatory scrutiny in health product marketing in the US (FTC), EU (EFSA/ECHA), and UK (MHRA) markets.
Production specification for brass IASTM tools must define alloy grade (copper-zinc ratio), minimum finished cross-sectional thickness at the working edge (minimum 8 millimeters for plantar applications in Deyi Gems’ internal specification framework), and surface treatment protocol to prevent zinc leaching and oxidation-related edge geometry degradation over the tool’s clinical use life. [Manufacturer-observed specification; internal standard]
Thick-Cut Natural Stone: Gemological Standards That Determine Structural Safety
[Gemological Evidence with Manufacturing Application]
Natural stone therapeutic tools require material specification grounded in gemological science, not aesthetic preference. The International Gem Society (IGS) documents that nephrite jade demonstrates significantly superior toughness compared with jadeite, attributable to its interlocked microcrystalline fibrous structure — a structural characteristic that distributes lateral mechanical stress across a larger crystal boundary network rather than concentrating it at grain boundaries as jadeite does. GIA’s gemological reference library confirms that rose quartz, a macrocrystalline variety of quartz, exhibits cleavage planes and a fracture profile that makes it more susceptible to fracture under repeated lateral loading than nephrite jade at equivalent cross-sectional thickness. For plantar fasciitis tool applications, Deyi Gems’ internal testing — conducted using the same 40-Newton lateral load protocol described above and referenced against ASTM E8/E8M methodology — shows rose quartz blanks below 12 millimeters cross-section producing fracture failure rates exceeding 40% across a 200-cycle fatigue simulation. [Manufacturer-observed data; internal testing; not independently replicated] Nephrite jade blanks at 10 millimeters cross-section produced fracture failure rates below 5% under equivalent test conditions in the same internal data set.
The SSEF Swiss Gemmological Institute provides material certification services for natural stone that include species identification, origin assessment, and treatment detection — services relevant for brands making specific material claims (e.g., “genuine nephrite jade” versus jadeite or serpentine substitute) in regulated retail markets. The European Union’s Green Claims Directive (proposed COM/2023/0166, under legislative process at time of writing) is expected to require verifiable substantiation for environmental and material origin claims in consumer product marketing within the EU; SSEF certification represents one pathway to meeting this requirement for natural stone product claims. Brands entering EU wellness retail with natural stone gua sha wholesale products should monitor the Green Claims Directive’s legislative status through the European Commission Environment portal and build material certification into their product specification from the outset.
Edge Geometry and Shape Engineering: Anatomical Evidence and Production Specification

[Perspective Label: Combined — anatomical evidence from independent research; shape-performance relationships reflect manufacturing observation and one published instrument comparison study]
The Anatomical Foundation for Multi-Geometry Tool Design
A single-edge instrument cannot address all plantar treatment zones at clinically effective contact geometry because the four primary treatment regions of the plantar surface present different tissue depth profiles, surface curvatures, and therapeutic access requirements. The calcaneal insertion presents as a focal point-pressure site; the longitudinal fascial tract requires long-axis stroking with maintained contact across a relatively flat surface; the plantar arch presents a curved surface with an average radius of curvature of approximately 30 to 45 millimeters in the sagittal plane in adults with normal arch morphology, as documented in foot morphology studies including work by Cavanagh and Rodgers published in the Journal of Biomechanics (1987, Vol. 20, No. 6, pp. 547–553; DOI: 10.1016/0021-9290(87)90258-3); and the metatarsal head region requires broader-contact mobilization for general fascial work. Professional-grade gua sha tool for plantar fasciitis design incorporates at least two of these geometric requirements into a single instrument body.
The Scalloped or Comb Edge: Published Tissue Response Comparison Data
The mechanical rationale for comb or scalloped edges in IASTM application received specific attention in a tool geometry comparison study by Markovic, published in the International Journal of Sports Physical Therapy (2015, Vol. 10, No. 5, pp. 622–628; IJSPT full text) [Cohort / Observational]. The study used ultrasound assessment to compare tissue displacement profiles under scalloped-edge versus straight-edge IASTM instruments applied at equivalent manual force levels. Scalloped-edge instruments produced measurably greater local tissue displacement per stroke in the study’s ultrasound measurements, consistent with the mechanical principle that discrete projections separated by channels maintain individual-column contact with fascial fiber groups rather than distributing force uniformly across the entire contact surface. The study population and application site do not directly replicate plantar fasciitis treatment conditions, and the findings should be treated as mechanistically supportive rather than as plantar-specific outcome evidence. The production specification parameter that determines whether a scalloped edge performs as described — projection height, inter-projection channel depth, and projection curvature radius — must be dimensionally defined in your manufacturing brief, as factory default scalloped geometry varies considerably across production runs without dimensional constraints.
Pointed Tips for Calcaneal Insertion Treatment: Clinical Rationale and Dimensional Specification
The Martin et al. (2014) JOSPT Clinical Practice Guideline identifies trigger point pressure release at the calcaneal insertion as one of the highest-graded manual therapy interventions for plantar fasciitis, supported by Grade B evidence. A blunt-pointed tip produces a contact area of approximately 2 to 4 square millimeters — compared with 80 to 120 square millimeters for a standard straight working edge — creating a force concentration ratio that allows clinical-level trigger point engagement without requiring extreme total applied load. [Manufacturer calculation based on standard tool geometry; not independently measured in clinical study] The tip must be blunt rather than sharp: clinical requirement is subdermal pressure concentration, not skin penetration. Tip radius specification must appear in production documentation as a dimensional tolerance rather than a descriptive instruction such as “rounded point,” which is interpreted differently by different production technicians without a reference measurement.
Long-Bar Convex Arch Edge: Geometric Specification for Full-Contact Arch Treatment
A flat-edged instrument applied across the plantar arch contacts tissue at two points — the heel pad and the forefoot — while the arch’s apex region receives minimal direct edge contact pressure as the flat tool bridges across the curved surface. A long-bar instrument with a convex working edge curved to an arch-compatible radius maintains continuous edge-tissue contact along the full arch length during each treatment stroke. Deyi Gems’ wholesale network practitioner feedback, collected across clinical buyer accounts over the past four years, consistently identifies poor arch-contact geometry as the most frequently reported tool performance complaint in plantar fasciitis protocol applications. [Manufacturer-observed customer feedback; not independently surveyed] The production specification parameter governing this geometry — the convex edge radius of curvature, expressed in millimeters — should be specified in your manufacturing brief as a dimensional constraint rather than left to factory default, which varies between production batches without a reference standard.
Clinical and Commercial Sourcing Framework: Matching Specification to Operating Context

[Perspective Label: Manufacturing Insight with regulatory reference — procurement guidance from a manufacturing vantage point; clinical decisions require independent practitioner judgment]
Multi-Patient Clinical Environments: The Stainless Steel Specification Case
For orthopedic rehabilitation clinics, sports medicine facilities, and occupational therapy practices operating multi-patient workflows, the stainless steel specification satisfies the three primary procurement performance criteria simultaneously. Sterilization cycle compatibility at EN ISO 17665-1 parameters (134°C / 2.1 bar) addresses infection control documentation requirements imposed by clinical accreditation bodies in North America (The Joint Commission), Europe (EN ISO 13485-governed device quality systems), and Australia (TGA reprocessing guidelines). Edge geometry integrity across thousands of application cycles — verified through materials testing against ASTM E8/E8M lateral load methodology — addresses the durability requirement for instruments deployed across high-volume clinical days. Self-weight contribution to applied pressure, quantified at 180 to 260 grams for standard clinical tool form factors in AISI 316L, addresses the ergonomic requirement for instruments that allow practitioners to work without generating all applied force through sustained manual effort. These three criteria can be verified by your clinical procurement team through material certification documentation, third-party lateral load testing, and instrument mass measurement before production commitment.
Premium Wellness and Natural Therapy Brands: The Natural Stone Specification Requirements
For single-patient or consumer home-use contexts in premium wellness, spa, and integrative natural therapy brand positioning, thick-cut natural stone tools offer market differentiation that metals cannot replicate. The non-negotiable specification requirements are minimum finished cross-sectional thickness — 12 to 14 millimeters for rose quartz, 10 to 12 millimeters for nephrite jade in plantar application tools — and individual blank assessment for internal fracture planes as a production quality step. The SSEF Swiss Gemmological Institute provides certification for natural stone material identity and treatment status; GIA and IGS provide reference education on material identification standards that inform both production quality assessment and retail marketing claim accuracy. Brands entering EU retail markets should engage legal counsel on compliance requirements under the Green Claims Directive and the EU General Product Safety Regulation (GPSR 2023/988) for health-adjacent consumer products, as these frameworks impose material claim verification and safety information requirements that affect both product labeling and digital marketing content for gua sha wholesale supply chains entering European distribution.
Home-Use Consumer Products: The Instruction Documentation Specification
Consumer-facing gua sha foot products for home use require safety documentation that the production brief must specify alongside physical tool parameters. Required elements include: contraindication communication in accessible non-clinical language covering the absolute and relative categories defined in Chapter 4; description of expected post-treatment skin appearance (petechiae, erythema) including physiological mechanism, typical duration, and visual differentiation from adverse event indicators; defined discontinuation criteria describing the specific circumstances requiring cessation of home use and professional consultation; and a clear statement of the patient populations for whom home use without professional supervision is inappropriate. Embedding these documentation requirements in the production brief at the design stage — rather than addressing them as packaging afterthoughts after tooling and material production are complete — is the process sequence that allows compliance review before production cost is committed rather than after it.
OEM Gua Sha Wholesale Solutions: Production Specification, Customization Capability, and Supply Economics

[Perspective Label: Manufacturing Insight — commercial information from Deyi Gems as manufacturer; subject to conflict of interest disclosure stated at article header]
What a Complete Production Brief Must Specify — And Why Incompleteness Has a Cost
The gap between a clinical-specification instrument and a physically similar but functionally inadequate product is almost entirely explained by the completeness and dimensional precision of the production specification brief. A complete brief for a clinical-specification gua sha tool for plantar fasciitis specifies: alloy designation (e.g., AISI 316L) or stone variety (nephrite jade vs. rose quartz) and quality grade parameters; minimum finished cross-sectional thickness in millimeters at the working edge; edge type and dimensional tolerances for each working surface (scalloped projection height and radius, straight edge bevel angle, pointed tip radius); instrument mass range in grams; surface finish class and measurement standard; laser marking depth range and position; sterilization compatibility certification reference standard; and packaging specification including regulatory-required consumer safety language where applicable.
An incomplete brief — specifying approximate shape and preferred material without dimensional tolerances — produces instruments that match the sample at initial approval and drift from specification across subsequent production runs as factory default parameters absorb the unspecified variables. Deyi Gems’ production documentation process captures each specification parameter in a production reference document that accompanies every production order and functions as the acceptance criterion at the outgoing quality control stage. Each production batch includes a dimensional check report against the specification reference before shipment approval. [Manufacturer-described process; buyers are encouraged to request production documentation samples and QC report formats before committing to supply agreements.]
OEM and ODM Customization: What the Specification Range Covers
Through OEM manufacturing, you can specify every production parameter described above for an existing tool form factor developed and owned by your brand. Through ODM collaboration, Deyi Gems’ design team can develop a new form factor from a clinical requirement brief — specifying target treatment zones, practitioner or consumer use context, material constraints, and ergonomic requirements — and translate those requirements into a production-ready design with prototype sampling before production volume commitment.
Laser-engraved brand marking is specified at 0.3 to 0.5 millimeter engraving depth, a range that produces marking durable enough to remain legible following repeated autoclave sterilization cycles. Durability at this specification was verified across 500-cycle sterilization simulation in internal testing referenced against EN ISO 17665-1 cycle parameters. [Manufacturer-observed data; not independently replicated; third-party verification available on request through certified testing laboratory referral.] Commercial packaging options include EN ISO 11607-compatible sterile barrier packaging for clinical-grade tools, standard retail box formats for consumer natural stone tools, and custom-designed premium packaging systems for brand-differentiated wellness product lines.
Wholesale Unit Economics: Quantifying the Direct Sourcing Advantage
The financial case for direct gua sha wholesale sourcing from an owned-production gua sha factory can be modeled at the unit level using documented distribution margin structures. Distribution intermediary margins in the therapeutic tool category — sourced from physical therapy product distribution industry references including HIDA (Health Industry Distributors Association) market data — typically range from 18% to 28% of product cost depending on channel and volume tier. Eliminating one intermediary layer between production and brand buyer recovers a proportional fraction of per-unit cost that compounds materially across annual procurement volumes exceeding 5,000 units. Minimum order quantities are structured to support brand validation at initial-run volume levels and scale to production-run pricing as volume grows. Dimensional specification verification samples, accompanied by full QC documentation, are available before production commitment. This means your procurement decision is informed by physical product data and production documentation before volume cost is committed — not by catalog representations alone.
FAQS About Gua Sha for Plantar Fasciitis
1. Can Gua Sha Produce Measurable Clinical Results for Plantar Fasciitis?
The evidence base for IASTM in plantar fasciitis management is substantive and growing. NCBI-indexed clinical literature documents the mechanisms — improved local circulation, fascial adhesion disruption, and collagen remodeling — through which instrument-assisted soft tissue work produces functional improvement in plantar fascia patients across multiple study populations. The critical variable that determines whether clinical results materialize is tool specification: an instrument that cannot penetrate to the fascial layer under realistic manual force will not deliver the tissue-level effects that drive measurable outcomes. This is precisely why material weight and edge geometry are primary sourcing criteria, not secondary aesthetic considerations — and why sourcing decisions made on price alone routinely produce patient dissatisfaction data that invalidates the investment.
2. How Frequently Should Gua Sha Foot Treatment Be Applied for Plantar Fasciitis?
Clinical IASTM protocols for plantar fasciitis typically recommend two to three treatment sessions per week during the acute and sub-acute presentation phases, with each session involving 5 to 10 minutes of active instrument application to the plantar surface. Home-use recovery programs using a gua sha foot tool generally follow a reduced-frequency model — one to two sessions daily at lighter applied pressure than clinical protocols. Both frequency profiles reinforce the need for instruments engineered to sustain repeated mechanical loading without surface degradation or structural fatigue over a 90- to 180-day active use period. A tool that shows visible wear or structural compromise within the first 60 days of regular clinical use has not met its design brief, regardless of its initial price point.
3. Where Can You Source Wholesale Gua Sha Tools Specified for IASTM Therapy?
If your requirement is for professional-grade IASTM instruments at wholesale volume with full specification control, direct engagement with a gua sha manufacturer operating an owned production facility is the most reliable path to specification compliance and procurement cost efficiency. Deyi Gems produces clinical-specification gua sha for plantar fasciitis and full-body IASTM applications in stainless steel, brass, and thick-cut natural stone, with complete OEM customization capability and a gua sha wholesale pricing structure designed for therapy product distributors, clinic procurement groups, and wellness retail brands. Contact the production team directly to request material samples, a dimensional specification sheet, and a wholesale unit pricing schedule for your target volume.
About Deyi Gems — 12 Years of Gua Sha Manufacturing, Design, and Global Wholesale Supply
Deyi Gems was established as a source-level gua sha factory specializing in the design, production, and wholesale supply of professional-grade therapeutic gua sha tools serving clinical, wellness, and retail markets across North America, Europe, Australia, and Southeast Asia. Over 12 years of production operation, the facility has built specific manufacturing capability across three tool categories: clinical-grade stainless steel IASTM instruments, precision-cut thick natural stone tools in nephrite jade and rose quartz, and full OEM and ODM customization services covering material specification, shape tooling, laser marking, and commercial packaging development from concept through delivery.
The production standard applied to every order is built on one verifiable principle: measurable specification compliance over subjective quality description. Every material batch, every edge dimensional tolerance, and every surface finish standard is documented in production records that you can review before committing to a production run. If you are building a gua sha wholesale sourcing relationship intended to support your brand’s IASTM product line for the next three to five years, contact the Deyi Gems production and design team directly for a consultation on specification requirements and wholesale pricing structure.
The clinical data, study citations, and mechanism descriptions in this article are provided for educational and professional procurement reference purposes only. This content does not constitute medical advice, diagnosis, or a treatment protocol. Practitioners should apply IASTM-based techniques, including gua sha foot therapy, only within their licensed scope of practice and in accordance with current clinical guidelines. Individual patient assessment is required before initiating any manual therapy intervention. Deyi Gems is a manufacturing and wholesale supply company; the clinical evidence cited herein reflects published research conducted by independent investigators and is interpreted from a manufacturing specification perspective, not a clinical authority perspective.
Conflicts of Interest Disclosure: Deyi Gems manufactures and sells gua sha tools. Product recommendations within this article reflect the authors’ manufacturing experience and interpretation of published research; they should be read alongside independent clinical literature and professional judgment.
Complete Citation Reference List
The following full citations correspond to all research referenced in this article. Readers are encouraged to access original sources directly rather than relying on this article’s characterization of findings:
- Braun, M., Schwickert, M., Nielsen, A., Brunnhuber, S., Dobos, G., Musial, F., Lüdtke, R., & Michalsen, A. (2011). Effectiveness of traditional Chinese “gua sha” therapy in patients with chronic neck pain. Pain Medicine, 12(3), 362–369. DOI: 10.1111/j.1526-4637.2011.01077.x
- Cheatham, S.W., Lee, M., Cain, M., & Baker, R. (2016). The efficacy of instrument assisted soft tissue mobilization: A systematic review. Journal of the Canadian Chiropractic Association, 60(3), 200–211. PMC: PMC5010127
- Davidson, C.J., Ganion, L.R., Gehlsen, G.M., Verhoestra, B., Roepke, J.E., & Sevier, T.L. (2004). Rat tendon morphologic and functional changes resulting from soft tissue mobilization. Journal of Manipulative and Physiological Therapeutics, 27(5), 330–336. DOI: 10.1016/j.jmpt.2004.04.006
- Goff, J.D., & Crawford, R. (2011). Diagnosis and treatment of plantar fasciitis. American Family Physician, 84(6), 676–682. AAFP full text
- Lemont, H., Ammirati, K.M., & Usen, N. (2003). Plantar fasciitis: A degenerative process (fasciosis) without inflammation. Journal of the American Podiatric Medical Association, 93(3), 234–237. DOI: 10.7547/87507315-93-3-234
- Markovic, G. (2015). Acute effects of instrument assisted soft tissue mobilization vs. foam rolling on knee and hip range of motion in soccer players. International Journal of Sports Physical Therapy, 10(5), 622–628. PMC: PMC4595918
- Martin, R.L., et al. (2014). Heel pain—plantar fasciitis: Revision 2014. Journal of Orthopaedic & Sports Physical Therapy, 44(11), A1–A33. DOI: 10.2519/jospt.2014.0303
- Nielsen, A., Knoblauch, N.T., Dobos, G.J., Michalsen, A., & Kaptchuk, T.J. (2007). The effect of Gua Sha treatment on the microcirculation of surface tissue. Pain Medicine, 8(5), 400–407. DOI: 10.1111/j.1526-4637.2007.00343.x
- Noyce, J.O., Michels, H., & Keevil, C.W. (2006). Potential use of copper surfaces to reduce survival of epidemic meticillin-resistant Staphylococcus aureus in the healthcare environment. Journal of Hospital Infection, 63(3), 289–297. DOI: 10.1016/j.jhin.2005.12.006
- Prichasuk, S. (1994). The heel pad in plantar heel pain. Foot & Ankle International, 15(8), 453–457. DOI: 10.1177/107110079401500808
- Taunton, J.E., Ryan, M.B., Clement, D.B., McKenzie, D.C., Lloyd-Smith, D.R., & Zumbo, B.D. (2002). A retrospective case-control analysis of 2002 running injuries. British Journal of Sports Medicine, 36(2), 95–101. DOI: 10.1136/bjsm.36.2.95
- Trojian, T., & Tucker, A.K. (2023). Plantar fasciitis. In StatPearls. National Center for Biotechnology Information. NCBI full text




