
If you’ve ever paid $150 for a “medical-grade gua sha treatment” and walked away wondering why the results felt no different from a $60 spa session — you may have been overcharged, undertreated, and legally unprotected all at once. Is gua sha legit? The short answer is yes — the tool itself is completely legal across Asia, North America, and Europe. But the service behind it? That’s where the legal and physical risks begin. Consumers are routinely paying medical-tier prices for beauty-tier services, delivered by practitioners who hold no qualifying credentials. And in some cases, those same practitioners are using substandard tools that introduce skin and circulatory risks that a licensed provider would never allow. Before you book your next session — or place your next B2B order — here’s what the industry won’t tell you.
The Tool Is Legal. The Service Behind It May Not Be.
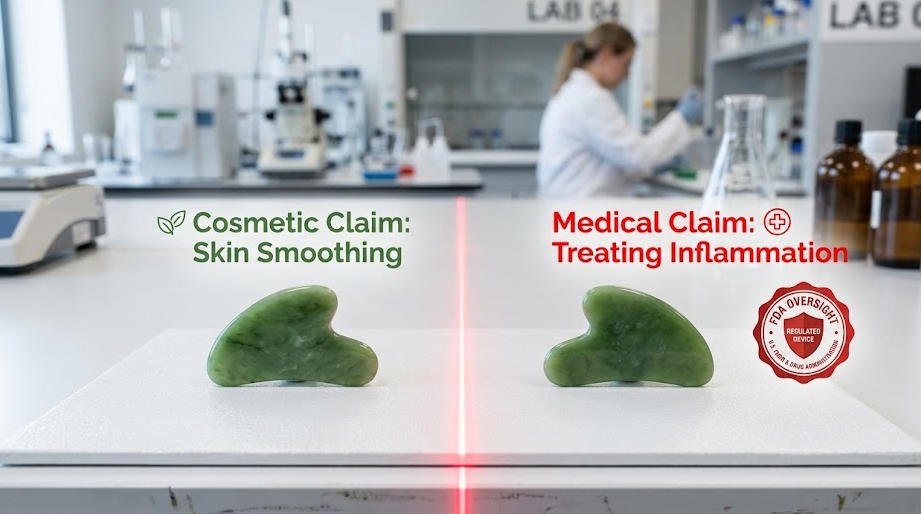
There is a persistent misconception in the wellness market: because gua sha tools are sold openly on Amazon, in spas, and through health retailers, the entire practice must fall under a single legal umbrella. It does not. Gua sha as a physical instrument — whether crafted from jade, rose quartz, or bian stone — is a legal consumer product in every major market it serves. Regulatory bodies in the United States, the European Union, and across Southeast Asia do not restrict the manufacture or general sale of gua sha tools. What they do regulate, often rigorously, is the context in which those tools are used and the claims made about what they can do.
Why the Tool Itself Is Not the Legal Variable
The legal status of a gua sha tool is fixed: it is a wellness or cosmetic product until someone changes its designation through the claims they make about it. A jade facial roller sold as a skincare accessory on a beauty website sits in an entirely different regulatory category than the same tool marketed as a device capable of “treating chronic inflammation” or “accelerating lymphatic drainage for diagnosed conditions.” The moment a seller, clinic, or practitioner attaches a medical claim to the tool or the service, an entirely different body of law applies. In the United States, this falls under FDA oversight for medical devices. In the EU, it triggers compliance requirements under the EU Medical Device Regulation (MDR 2017/745). Ignoring these thresholds is not a gray area — it is a prosecutable violation.
The Two Tracks That Define Gua Sha Legality
Every gua sha business, whether a manufacturing operation, a retail brand, or a treatment clinic, operates on one of two distinct legal tracks. The first is the beauty and general wellness track, which covers facial gua sha, relaxation massage, and daily self-care tools sold without medical claims. This category requires no special medical licensing, no clinical certification, and no government-issued healthcare registration — as long as the business itself is operating lawfully. The second is the medical or therapeutic track, which applies the moment a practitioner or brand claims that gua sha can treat, diagnose, or prevent any health condition. This track demands practitioner licensure, institutional accreditation, liability insurance calibrated for clinical services, and in some markets, product registration as a Class I or Class II medical device. Understanding which track your provider — or your product — sits on is the single most important question you can ask before spending money or accepting risk.
The $150 Price Gap — How Unlicensed Clinics Cost You More Than Money

One of the most financially damaging patterns in the gua sha market is also one of the least discussed. Consumers walk into clinics that advertise “medical-grade gua sha therapy,” pay accordingly, and leave having received nothing more than a standard beauty treatment delivered by someone without the training — or the legal authority — to provide anything more. This is not a rare edge case. Based on market surveys covering both in-person clinics and institutional pricing published on provider websites, the pricing gap between beauty-tier and medical-tier gua sha services is significant and consistent: beauty gua sha sessions typically run between $50 and $100 per visit, while sessions marketed as medical or therapeutic gua sha are priced between $80 and $200 per session. That gap exists for legitimate reasons. When those reasons are absent — when the clinic lacks credentials but keeps the premium price — you absorb the entire financial loss with no recourse.
Why Medical Gua Sha Costs More — and Why That Premium Is Justified
The price differential between beauty and medical gua sha is not arbitrary, and understanding its structure helps you identify when you’re being charged fairly versus when you’re being exploited. Medical-tier gua sha costs more because it is more — across four measurable dimensions. First, labor costs are substantially higher: licensed practitioners — whether they hold Traditional Chinese Medicine (TCM) certifications, acupuncture licenses, or physical therapy credentials — spend years acquiring qualifications that command higher compensation. Second, service depth differs meaningfully: a medical gua sha session involves intake assessment, contraindication screening, pressure calibration based on the patient’s condition, and post-treatment monitoring. A beauty session involves none of these steps. Third, session structure reflects the difference: medical gua sha is typically integrated into a multi-session treatment plan with documented progress tracking, whereas a beauty session is a standalone experience with no clinical continuity. Fourth, and perhaps most importantly, legal and liability costs are built into every medical-tier session: malpractice insurance, clinical compliance overhead, and regulatory filing requirements add real operational costs that licensed providers must absorb. When you pay $160 for a session, a meaningful portion of that is purchasing protection — yours and theirs.
The Trap: Medical Pricing, Beauty Execution
Here is where the financial risk becomes concrete. An unlicensed clinic that markets itself as offering “therapeutic” or “medical-grade” gua sha carries none of the four cost structures described above. They pay standard beauty labor rates, they perform standard beauty-depth service, they run no intake assessment, and they carry no clinical liability insurance — because they legally cannot. Yet their pricing reflects the medical tier because that positioning attracts clients willing to pay for perceived clinical value. The result is a transaction in which you pay $150–$200, receive a $50–$70 service, and have no legal basis for complaint because the provider will claim they never formally guaranteed medical outcomes. This is not technically fraud in the criminal sense in every jurisdiction — but it is a form of market deception that regulatory bodies in the U.S. and EU are increasingly scrutinizing. The Federal Trade Commission’s guidelines on deceptive health claims make clear that implying clinical efficacy without substantiation is an actionable violation, regardless of whether explicit guarantees were made.
What “Gua Sha Safe” Actually Costs to Deliver
When evaluating whether a provider’s price is justified, your reference point should not be the number itself but the infrastructure behind it. A gua sha service that is genuinely gua sha safe at the medical level requires documented practitioner credentials that you can verify independently, tools that meet material safety and hygiene standards, a client intake process that screens for contraindications such as blood clotting disorders or active skin infections, and post-session care protocols. Each of these elements has a cost. If a provider is charging you medical-tier prices but cannot demonstrate any of these infrastructural elements when asked, the price premium is not justified — and your financial and physical exposure is real. Asking for documentation before you pay is not rude. It is rational consumer behavior that every legitimate provider will accommodate without hesitation.
Is Gua Sha Safe? The Physical Risks Behind Unqualified Practice
Money lost to an unlicensed clinic is recoverable. Physical harm from an unqualified practitioner using substandard tools is not always so easily reversed. This is the dimension of the gua sha risks conversation that tends to be underemphasized in wellness marketing — because it is bad for business to discuss it openly. The reality is that gua sha, performed correctly by a trained individual using appropriate tools and lubricants, carries a very low risk profile. The National Center for Complementary and Integrative Health (NCCIH) acknowledges gua sha as a generally safe practice when administered properly, noting that the characteristic skin redness — known as sha — is an expected and temporary response, not an injury. But “when administered properly” is doing significant work in that sentence. Strip away the training, the tool quality, and the intake screening, and the risk profile changes in ways that are neither minor nor theoretical.
Known Gua Sha Risks Even Under Correct Conditions
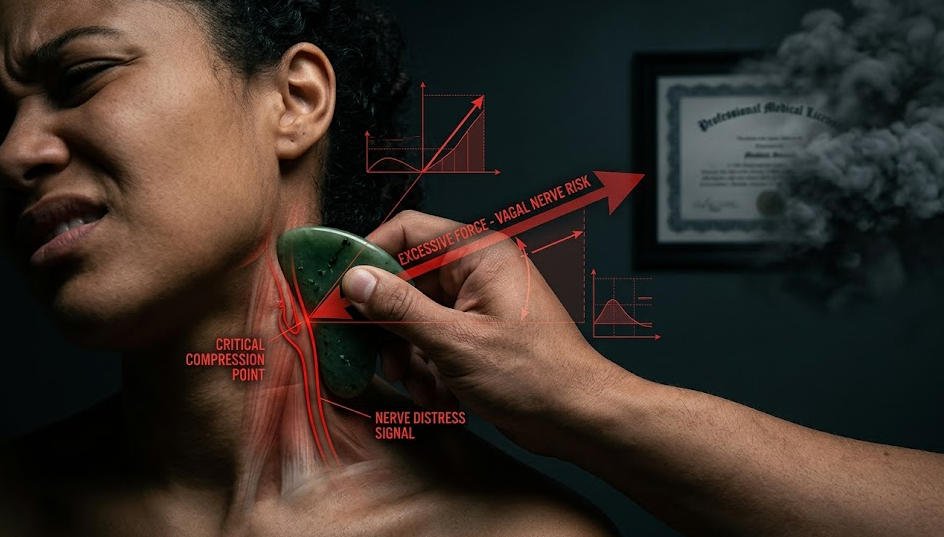
Before addressing what unqualified practice adds to the risk equation, it is worth being honest about what risks exist even under ideal conditions. Gua sha involves applying firm, directional pressure to the skin using a smooth-edged tool, and this process routinely produces petechiae — small broken capillaries beneath the skin surface that appear as redness or bruising. In a properly conducted session, this response is controlled, expected, and resolves within two to four days without intervention. However, individuals with certain conditions — including those taking anticoagulant medications, those with active inflammatory skin conditions, or those with undiagnosed bleeding disorders — face amplified responses that can escalate beyond cosmetic redness into genuine dermal injury. Additionally, vigorous gua sha applied to the neck and chest area has been associated in clinical case reports with transient increases in heart rate and, in rare instances, vagal nerve stimulation. These are not reasons to avoid gua sha. They are reasons why a competent practitioner screens clients before beginning, and why regulatory frameworks require medical-tier providers to carry the credentials that enable that screening.
How Unlicensed Practitioners Multiply Your Physical Exposure
An unlicensed practitioner operating without clinical training does not simply lack a piece of paper. They lack the diagnostic knowledge that the paper represents. They cannot reliably identify contraindications during a client intake. They have no protocol for responding to an adverse reaction mid-session. They are not covered by clinical malpractice insurance, which means that if harm occurs, your legal recourse is limited and the path to compensation is uncertain. Beyond the knowledge gap, there is a pressure calibration problem that is consistently underestimated. Medical-tier gua sha involves pressure levels, stroke directions, and treatment durations that are calibrated to the individual client’s condition, tissue density, and therapeutic goal. Beauty-tier gua sha applies generalized, light-to-medium pressure designed to promote circulation and relaxation without clinical depth. An untrained practitioner attempting to replicate medical-technique gua sha without the underlying training is not delivering a slightly inferior version of the same service — they are performing a categorically different and potentially harmful procedure. This is precisely why the licensing requirements exist, and why their absence is a physical risk factor, not merely an administrative irregularity.
The Low-Quality Tool Problem — A Risk Factor You Cannot See
There is a third layer of physical risk that operates entirely below the consumer’s awareness: the quality of the gua sha tool itself. Not all gua sha tools are manufactured to the same standards, and the differences that matter most are not visible to the naked eye. A properly manufactured jade or rose quartz gua sha tool should have edges that are smooth and uniformly radiused — neither so sharp that they cut the skin under pressure, nor so blunt that they require excessive force to generate therapeutic friction. Tools produced without quality control standards may have micro-fractures in the stone surface, inconsistent edge profiles, or surface finishes that create abrasion rather than glide. When combined with substandard lubricants — low-grade mineral oils or synthetic substitutes that contain irritants — the contact surface between tool and skin becomes a source of localized chemical and mechanical irritation that compounds over a session.
This is an area where the sourcing decisions made at the gua sha manufacturer level have direct downstream consequences for the person lying on the treatment table. A gua sha factory that applies material grading standards, edge-finishing protocols, and lubricant compatibility testing produces tools that behave predictably under clinical pressure. One that does not introduces variables that neither the practitioner nor the client can control. According to material quality frameworks referenced by the Gemological Institute of America (GIA), the structural integrity and surface characteristics of stones like nephrite jade and xiuyan jade vary significantly by origin and processing method — variations that translate directly into performance and safety differences at the tool level. You may not be able to inspect a tool’s internal grain structure before your session, but you can ask your provider where their tools are sourced and whether their supplier operates under documented quality standards. A provider who cannot answer that question is giving you information — just not the kind they intended.
What “Gua Sha Safe” Requires at Every Level of the Supply Chain
Genuine safety in a gua sha session is not the product of a single decision — it is the cumulative result of quality decisions made at every stage between raw material and skin contact. Your safety depends on a practitioner who is trained to the level their service claims. It depends on a clinic that operates legally and carries appropriate liability coverage. And it depends on tools sourced from a gua sha manufacturer that treats material integrity and edge quality as non-negotiable production standards rather than optional finishing steps. When any one of these links is missing, the risk does not remain contained to that link — it propagates forward to you. Understanding this chain of accountability is what separates an informed consumer from one who is simply hoping for the best.
The Legal Line Explained — Beauty vs. Medical Gua Sha Across Key Markets

Understanding whether a gua sha service is operating legally requires more than a general sense that “wellness practices are usually fine.” The legal frameworks governing gua sha vary by country, by intended use, and by the specific claims attached to the service — and the distance between a compliant business and a prosecutable one can be as narrow as a single sentence in a marketing brochure. Is gua sha legit as a commercial practice? In the beauty and general wellness category, almost universally yes. In the medical and therapeutic category, only when the provider has satisfied a defined set of regulatory requirements that differ meaningfully across jurisdictions. What follows is a practical breakdown of how those requirements operate in the markets where gua sha is most commercially active.
When Gua Sha Is Completely Legal — The Beauty and Wellness Category
For the vast majority of gua sha businesses — retail tool sellers, beauty spas, facial treatment studios, and general wellness centers — the legal requirements are straightforward and manageable. In the United States, a spa or massage studio offering gua sha as part of a relaxation or skincare service does not require FDA clearance for its tools, does not need practitioners to hold medical licenses, and does not face clinical liability standards, provided the service makes no medical claims. The business must hold standard local operating licenses, comply with general hygiene and safety codes applicable to personal care establishments, and ensure that its staff hold whatever cosmetology or massage therapy credentials are required by state law — which vary but are consistently achievable outside the medical licensing system. In the European Union, the same general principle applies: beauty-tier gua sha is treated as a personal care service and falls under consumer protection and general business licensing frameworks rather than medical device or healthcare provider regulations. Across Southeast Asia, where gua sha has the longest cultural history and the deepest market penetration, beauty-tier practice is similarly unencumbered by clinical licensing requirements in most jurisdictions, though specific rules vary by country.
When Gua Sha Becomes a Legal Risk — The Medical Claims Category
The legal environment changes fundamentally the moment a gua sha provider moves beyond beauty positioning and into therapeutic or medical claim territory. In the United States, any claim that a gua sha service can treat, diagnose, cure, or prevent a specific medical condition — including statements about managing chronic pain, accelerating recovery from injury, or improving diagnosed circulatory conditions — brings the practice into scope for both FDA medical device regulation and state healthcare practice laws. Practitioners making such claims without holding appropriate licensure — which may include acupuncture licenses, physical therapy credentials, or Traditional Chinese Medicine (TCM) practitioner registration depending on the state — are operating outside their legal scope of practice. The consequences are not trivial: unlicensed practice of a healthcare profession is a criminal offense in most U.S. states, carrying penalties that range from substantial fines to prosecution. The World Health Organization’s guidelines on traditional medicine practice reinforce the position that traditional therapeutic modalities, including gua sha, require formal practitioner competency frameworks when deployed in clinical or quasi-clinical contexts.
How the EU Approaches Medical Gua Sha Regulation
In the European Union, the regulatory threshold for medical-tier gua sha is governed by two intersecting frameworks. The EU Medical Device Regulation (MDR) 2017/745, which came into full effect in May 2021, establishes that any device — including a gua sha tool — that is intended by its manufacturer to be used for a medical purpose must be classified, registered, and CE-marked as a medical device before it can be legally placed on the market with medical claims. A jade gua sha tool sold as a beauty accessory requires no CE medical marking. The same tool sold with claims about treating musculoskeletal pain or improving diagnosed lymphatic conditions requires full MDR compliance, including clinical evaluation documentation and post-market surveillance planning. Simultaneously, practitioners delivering gua sha in a medical context within EU member states must comply with national healthcare professional licensing requirements, which — while varying in specifics across member states — universally require demonstrable clinical training, professional registration, and liability insurance calibrated for healthcare practice. For any gua sha manufacturer or retailer exporting into the EU market, understanding the MDR classification threshold for their product claims is not optional — it is a prerequisite for legal market access.
A Practical Compliance Checklist for Consumers
Knowing the legal framework in abstract terms is useful. Knowing how to apply it before you book a session is more useful. When evaluating a gua sha service provider — particularly one that markets itself as offering therapeutic or medical-grade treatment — there are three categories of verification that every informed consumer should pursue before payment. First, practitioner credentials: ask the provider directly which license or certification they hold, which regulatory body issued it, and how you can verify it independently. In the U.S., most state licensing boards maintain publicly searchable online databases where practitioner licenses can be confirmed in minutes. Second, tool sourcing and quality standards: ask where the tools used in your session are sourced and whether the supplier is a certified gua sha factory operating under documented quality control protocols. A provider who sources tools from a traceable, standards-compliant gua sha manufacturer is giving you a meaningful safety signal. Third, liability coverage: ask whether the clinic carries professional liability insurance for the services they are performing. A licensed medical practitioner is required to carry malpractice coverage. A beauty practitioner is typically covered under general business liability. An unlicensed provider claiming medical capability often carries neither — which means your recourse if something goes wrong is effectively zero.
Why These Questions Make Legitimate Providers More Confident, Not Less
A provider who is operating legally, credentialed appropriately, and sourcing tools responsibly will not be unsettled by any of the questions above. They will answer them readily, often before you finish asking, because documentation and transparency are built into their operational infrastructure. The provider who responds to these questions with vagueness, deflection, or irritation is giving you the most important answer of all — and it is telling you to walk away. The legal frameworks governing gua sha across major markets exist precisely because the stakes involved in unqualified practice are real: financial loss, physical harm, and a complete absence of institutional recourse. Treating compliance verification as a standard step in your consumer decision process — not an act of suspicion — is the most effective protection you have.
If You’re a Gua Sha Seller — What You Must Know Before Making Any Claims

The legal and safety risks described in the preceding chapters are not exclusively a consumer problem. If you are a gua sha retailer, a wellness brand building a product line, or a distributor supplying clinics and spas across international markets, the compliance decisions you make — and the product claims you attach to your inventory — determine whether your business operates securely or exposes itself to regulatory action, platform delisting, and reputational collapse. Is gua sha legit as a commercial product category? Unambiguously yes. But the legitimacy of your specific business within that category depends entirely on the alignment between what you sell, what you say about it, and what your regulatory standing actually permits you to say. This chapter is written for sellers, brands, and distributors who want to understand that alignment clearly before it becomes a problem.
Selling Gua Sha as a Beauty Product — Your Compliance Boundaries
If your business sells gua sha tools as beauty or skincare accessories — facial rollers, body contouring tools, spa kits — your compliance obligations are relatively contained and achievable without significant legal infrastructure. In this category, your product claims should remain within the realm of cosmetic and wellness benefit: improved skin appearance, enhanced relaxation, improved product absorption, and general circulation support are claim territories that do not trigger medical device regulation in most major markets. What you must avoid, regardless of how compelling it might be from a marketing perspective, is any language that implies your product can treat, diagnose, prevent, or cure a physical condition. Phrases such as “clinically proven,” “treats inflammation,” “repairs tissue damage,” or “therapeutic-grade for medical use” move your product out of the beauty category and into medical device territory — with all of the regulatory obligations that entails — regardless of your intent. The FDA’s guidance on distinguishing cosmetic from medical device claims provides a practical reference for U.S.-market sellers navigating this boundary, and the principles it establishes translate broadly to EU and Asian market frameworks as well.
The Practical Claim Audit Your Product Line Needs Right Now
Every gua sha seller operating at scale should conduct a systematic audit of their current product claims — across their website, Amazon or third-party platform listings, social media content, influencer briefs, and packaging copy — specifically looking for language that crosses from wellness positioning into medical claim territory. This is not a theoretical risk management exercise. Major e-commerce platforms including Amazon have increasingly enforced health claim policies that result in listing suppression or account suspension without prior warning. Your marketing team’s enthusiasm for strong benefit language is understandable, but the business consequences of an unchecked medical claim on a product listing are measurably worse than the conversion lift that strong language might generate. A clean, compliant product claim architecture does not limit what your product does — it limits what you are permitted to assert about what it does. That is a legal boundary, not a marketing constraint, and treating it as such protects your revenue continuity.
Selling Gua Sha as a Medical Device — Certifications You Actually Need
If your business model involves selling gua sha tools with therapeutic or medical positioning — to clinics, licensed practitioners, hospital wellness departments, or directly to consumers with medical-benefit claims — your compliance requirements expand substantially, and the cost of non-compliance rises in proportion. In the United States, a gua sha tool marketed for medical therapeutic use must be evaluated under FDA medical device classification standards. Depending on the specific claims and intended use, this may require a 510(k) premarket notification submission demonstrating that your device is substantially equivalent to a legally marketed predicate device, or in some cases a full Premarket Approval (PMA) process. In the European Union, as previously noted, MDR 2017/745 compliance is mandatory for medically claimed devices, including clinical evaluation documentation, technical file preparation, and engagement with a Notified Body for conformity assessment. In markets across Southeast Asia — including Singapore, South Korea, and Japan — equivalent national medical device registration frameworks apply, each with their own submission requirements and review timelines.
The certification investment required to operate legitimately in the medical gua sha category is real, and it is not recoverable through shortcuts. However, the market access and pricing authority that certified medical-tier positioning provides are also real: as the pricing data earlier in this article establishes, the per-session revenue premium in the medical category is 60–100% above beauty-tier pricing, and the B2B wholesale relationships available to certified medical device suppliers operate at margins and order volumes that the beauty wellness category cannot match. For a gua sha manufacturer or brand evaluating whether to pursue medical certification, the question is not whether the investment is large — it is whether the market opportunity justifies the investment, and in most cases, the answer for a well-positioned supplier is yes.
Why Your Supplier’s Credentials Affect Your Legal Exposure
There is a dimension of seller compliance that is consistently overlooked by brands and distributors who focus exclusively on their own claims and certifications: the regulatory standing of your upstream gua sha manufacturer or gua sha factory has direct implications for your own legal position. If you are sourcing tools from a manufacturer that cannot provide documentation of their material sourcing, production quality controls, and — where applicable — product registration or safety testing, you are building your compliance architecture on an undocumented foundation. In a product liability scenario, your ability to demonstrate due diligence in supplier selection is a meaningful factor in determining your exposure. Regulatory bodies in the U.S. and EU increasingly hold importers and brand owners responsible for the compliance of products they place on the market, regardless of where those products were manufactured.
This means that when you evaluate a gua sha factory as a potential supplier, the questions you should be asking go beyond unit price, minimum order quantity, and lead time. You should be asking for material certifications — particularly for stone materials, where origin, treatment history, and structural integrity all affect product safety and performance. The International Gem Society (IGS) provides accessible reference standards for jade and related materials that responsible manufacturers should be able to cross-reference against their sourcing documentation. You should be asking for edge-finishing and quality control process documentation. And if your intended market is the EU, you should be asking whether the manufacturer has experience supporting MDR technical file preparation — because that experience reflects a level of production discipline and documentation rigor that translates into reduced compliance risk for your brand. Choosing a supplier on price alone when your product will carry health-adjacent claims is a risk calculation that rarely ends favorably.
The Business Case for Getting Compliance Right From the Start
Sellers who treat compliance as a cost center consistently underestimate the revenue consequences of getting it wrong. A product listing suspended for an unauthorized medical claim does not just lose its ranking — it loses its review history, its conversion rate data, and its visibility in a competitive market that is expensive to re-enter. A distributor whose supplier cannot provide material documentation faces the risk of a customs hold or product recall that disrupts not just one SKU but an entire import relationship. A brand that builds its identity around medical-tier gua sha positioning without the certification infrastructure to support that positioning is one regulatory inquiry away from a reputational event that no amount of marketing can reverse. By contrast, a seller who invests in compliant claim architecture, sources from a documented gua sha manufacturer, and pursues appropriate certification for their intended market creates a business that can scale into institutional and clinical channels — the highest-margin, highest-loyalty segment in the entire gua sha market. The compliance investment does not reduce your commercial opportunity. It defines the ceiling of how large that opportunity can grow.
Why We Know This — A Gua Sha Manufacturer’s Insider Perspective

Most articles about gua sha safety and legality are written from the outside looking in — by wellness journalists, legal commentators, or consumer advocates who understand the regulatory landscape in the abstract but have never sourced a single stone, negotiated a quality control protocol, or navigated a medical device certification process for a physical product. This article is written from a different position. Deyi Gems has operated as a source-level gua sha manufacturer for over 12 years, supplying international wellness brands, clinical distributors, and retail partners across North America, Europe, and Asia with jade and gemstone gua sha tools that are produced under documented material and finishing standards. That operational history — and specifically the experience of working across both the beauty wellness category and the emerging medical-grade gua sha segment — is what makes the distinctions drawn throughout this article something other than theoretical. What follows is not a company biography. It is an explanation of why direct manufacturing experience produces insights that no amount of secondary research can fully replicate.
What 12 Years of Manufacturing Reveals About the Tool Quality Problem
When you operate a gua sha factory at source level — meaning you are selecting raw stone material, overseeing cutting and shaping processes, managing edge-finishing quality control, and formulating or approving lubricant compatibility — you develop an understanding of tool quality variation that is invisible to anyone downstream in the supply chain. The difference between a nephrite jade gua sha tool that performs safely under clinical pressure and one that introduces micro-abrasion risk at the skin surface is not a difference you can see in a product photograph or detect from a wholesale price list. It is a difference in raw material grade, in the precision of edge radiusing during finishing, and in the surface polish depth achieved before the tool leaves the production floor. Over 12 years of production, Deyi Gems has refined the material grading and finishing standards that allow tools to perform consistently — meaning you receive a tool whose edge behavior, surface friction coefficient, and structural integrity are predictable across every unit in a production run, not just the samples submitted for approval.
This matters to you as a buyer or brand for a reason that goes beyond quality assurance in the abstract. When your tools perform consistently, your practitioners or your customers can develop reliable technique and pressure calibration. When tools vary — as they do from suppliers who do not apply consistent grading and finishing standards — the risk of unintended skin response increases with every session. A gua sha safe treatment experience is partly a function of practitioner skill and partly a function of whether the tool in their hand behaves the way they expect it to. You deserve both, and sourcing from a manufacturer who can document their production standards is how you secure the second half of that equation.
The Medical-Grade Development Process — And What It Taught Us About Compliance
In recent years, Deyi Gems has extended its product development work into the medical-grade gua sha segment — designing and testing tools intended for use by licensed practitioners in clinical and therapeutic settings. This process has involved direct engagement with medical device regulatory frameworks in multiple markets, collaboration with clinical practitioners to understand functional performance requirements that differ substantially from beauty-tier use cases, and material testing against standards that go beyond cosmetic safety into biocompatibility territory. The experience has been instructive in ways that informed every chapter of this article.
What the medical-grade development process makes undeniably clear is how large the gap is between a compliant medical gua sha tool and a standard beauty tool marketed with medical language. The compliance infrastructure required to legitimately position a gua sha tool for medical use — material biocompatibility documentation, clinical evaluation support, edge performance testing under therapeutic pressure loads, and in applicable markets, formal device registration — represents a genuine investment of both time and technical resources. A gua sha factory that has not engaged with this process cannot produce a tool that legitimately belongs in the medical category, regardless of what the product listing claims. And a brand that sources from such a factory and attaches medical claims to the product is building its market position on documentation that does not exist. This is why the distinction between beauty-tier and medical-tier gua sha is not a marketing nuance — it is a production reality that starts at the manufacturing level and propagates forward through every sale and every session.
Working With International Brands — What Compliance Conversations Actually Look Like
Deyi Gems works with wellness brands and distributors across multiple international markets, and the compliance conversations that accompany those relationships are consistently revealing about where the industry’s knowledge gaps are most pronounced. The most common misconception encountered in brand partner discussions is the belief that compliance is primarily a claims-language problem — that if you simply avoid the word “medical” in your product copy, you have addressed your regulatory exposure. This is an incomplete understanding of how product liability and regulatory enforcement actually operate. Your product’s positioning in the market — the imagery you use, the practitioners you feature in your marketing, the clinical language that appears in your customer reviews and goes unaddressed — all contribute to the regulatory picture that an enforcement body would evaluate if your product were flagged.
The second most common gap is in supplier due diligence. Brands that have invested seriously in their own compliance infrastructure — trademark registration, legal claim review, market-specific regulatory counsel — sometimes arrive at the supplier selection stage without applying equivalent rigor to their manufacturing partner evaluation. A brand’s compliance posture is only as strong as its weakest supply chain link, and a gua sha manufacturer who cannot provide traceable material documentation, production quality records, or regulatory filing support is a liability that sits below the surface of every product that brand brings to market. The brands that build the most defensible market positions are those that treat supplier compliance documentation as a non-negotiable component of their sourcing criteria — not a nice-to-have that gets addressed after the first purchase order.
What You Should Take Away From This Article
Whether you arrived at this article as a consumer wondering whether the gua sha session you just booked is being delivered by someone qualified to deliver it, or as a brand evaluating how to position your gua sha product line compliantly across multiple markets, the core insight is the same: is gua sha legit is a question with a fixed answer at the tool level and a variable answer at every level above it. The tool is legal. The practice is legal. What determines whether any specific instance of gua sha — as a service, as a product, as a business — sits on the right side of that legality is a combination of practitioner credentials, institutional licensing, product claim architecture, and manufacturing documentation that most participants in the market have never been asked to produce simultaneously.
You now know what to ask for. If you are a consumer, ask for practitioner credentials, tool sourcing documentation, and liability coverage confirmation before you pay for any service that carries therapeutic claims. If you are a seller or brand, audit your product claims against the regulatory frameworks of your target markets, source from a gua sha manufacturer whose production standards are documented and verifiable, and pursue the certifications that your intended market positioning actually requires rather than approximating compliance through careful word choice. And if you are evaluating manufacturing partners for a gua sha product line that you intend to scale into clinical or institutional channels — channels where the margin and loyalty dynamics are fundamentally superior to mass-market beauty retail — the production standards, regulatory experience, and material documentation that Deyi Gems has developed across 12 years of source-level manufacturing are directly relevant to the compliance foundation your brand needs.
A Final Word on the Industry’s Direction
The gua sha market is not standing still. Consumer sophistication about wellness product claims is increasing steadily, driven partly by high-profile enforcement actions against brands that overstated health benefits and partly by a growing base of consumers who have learned — sometimes through costly personal experience — to ask the questions this article has outlined. Regulatory bodies in the U.S., EU, and major Asian markets are dedicating more enforcement resources to the intersection of traditional wellness practices and medical claims, and the threshold for what constitutes an actionable claim is being interpreted more strictly with each passing year. The brands and practitioners that will hold durable market positions in this environment are those that built their compliance infrastructure before it was required of them — not those that are scrambling to retrofit documentation onto a product line that was constructed without it.
The gua sha risks that this article has documented — financial, physical, and legal — are not arguments against gua sha as a practice or as a product category. They are arguments for doing it correctly: with qualified practitioners, with tools manufactured to documented standards, with claims that reflect what the regulatory framework actually permits, and with the institutional transparency that turns consumer trust from a marketing aspiration into a verifiable, defensible fact. That is what legitimate gua sha looks like. And now you know exactly how to recognize it — and how to build it.
Faqs of Is Gua Sha Legit
1. Is gua sha legal to use and sell in the United States?
Yes — gua sha tools are legal consumer products in the United States. When sold and marketed as beauty or wellness accessories, they require no special regulatory clearance. The legal risk arises only when a seller or practitioner attaches medical or therapeutic claims to the tool or service without holding the appropriate FDA clearance, state practitioner license, or clinical certification. Keep your claims within cosmetic and general wellness territory, and your legal exposure remains minimal.
2. What are the most common gua sha risks consumers should know about?
When performed correctly, gua sha is a low-risk practice. The most common physical responses — skin redness and temporary surface bruising — are expected and resolve within a few days. The more serious risks emerge when sessions are delivered by unqualified practitioners who skip contraindication screening, apply incorrect pressure, or use substandard tools with uneven edges and low-grade lubricants. Consumers with blood-thinning medications, active skin conditions, or undiagnosed circulatory issues face amplified risk under unqualified hands.
3. How do I know if a gua sha provider is operating legally?
Ask three questions before you pay: What license or certification does your practitioner hold, and how can I verify it independently? Where are your tools sourced, and does your supplier operate under documented quality standards? Does your clinic carry professional liability insurance for the services being performed? A legitimately operating provider — whether beauty-tier or medical-tier — will answer all three questions without hesitation. Vagueness or deflection is your signal to walk away.
4. What is the difference between beauty gua sha and medical gua sha in terms of cost and service?
Beauty gua sha sessions typically cost between $50 and $100 per visit and focus on relaxation, skin appearance, and general circulation. Medical or therapeutic gua sha sessions are priced between $80 and $200 per session and involve clinical intake assessment, contraindication screening, condition-specific pressure calibration, and documented treatment planning. The price gap reflects real differences in labor, liability, and service depth — not marketing positioning. If a provider charges medical-tier prices without delivering medical-tier infrastructure, you are absorbing the financial loss directly.
5. Does the quality of the gua sha tool really affect safety?
Yes — more than most consumers realize. A properly manufactured gua sha tool has uniformly smooth, radiused edges that glide under pressure without creating skin abrasion. Tools produced without consistent quality control may carry micro-fractures, uneven edge profiles, or surface finishes that generate friction rather than therapeutic contact. When paired with low-grade lubricants, these variables compound into skin irritation and unpredictable session outcomes that neither the practitioner nor the client can anticipate. Sourcing tools from a documented gua sha manufacturer with traceable material standards and edge-finishing protocols is the most direct way to eliminate this risk from your sessions or your product line.



